Health
NAFDAC warns against using Covifor Remidesivir Injection
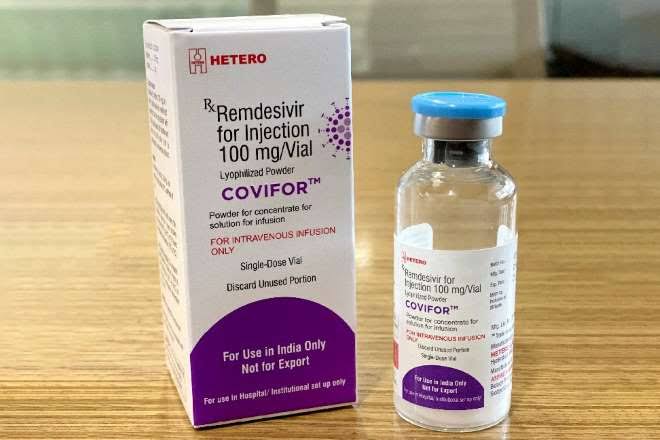
The National Agency for Food and Drug Administration and Control (NAFDAC) has warned the general public not to use ‘Covifor Remidesivir 100 mg Injection for treatment of any illness.
The warning was contained in a statement signed by the agency’s Director General, Prof. Mojisola Adeyeye, made available to the News Agency of Nigeria (NAN) on Friday in Abuja.
Remdesivir was originally developed by Gilead Sciences, a US-based pharma company, during the outbreak of Ebola to fight the deadly disease.
Remdesivir acts on the enzyme RNA polymerase which is necessary for the multiplication of the virus. When the virus could not make more copies of itself, it is not able to spread in the body and eventually dies. However, the drug didn’t seem to work as well as expected for the Ebola virus, thus was tested against the strains of coronavirus (SARS and MERS) in the year 2017 and now for SARS-CoV-2 in 2020.
The NAFDAC boss said that the label on the vaccine indicates that it was “Not for distribution in US, Canada or EU”, thus creating suspicion about its efficacy and could also have negative effect on the body system.
“The product is suspicious, and it could possibly be a falsified product, the general public is hereby warned to desist from the use of such product,” Adeyeye stressed.






